Introduction to this article:
February 1st, 2021
Dr. Robert Gorter wants to explain in as simple words as possible, the benefits and the risks of direct delivery of medications and nanoparticles through the nose into the brain.
- The brain is -to a certain extend- protected by the Blood-Brain barrier (BBB) which means that many molecules and substances that are toxic for the brain, are kept outside of the central nervous system.
- The skull is very thin between the roof of the nose and the brain (ca. 5 mm) and there are two options to deliver medications of nanoparticles (chips?) into the brain: a) by the thousands of olfactory nerve fibers and b) by nanoparticles which enter the brain like a “horse of Troy.”
- We are witnessing in current science in a rush in developing nanoparticles to deliver to / into the brain: one can wonder why?
- Some liposomes are developed as nanoparticles to deliver medications that would otherwise have been filtered out and stayed in the blood circulation to be detoxified in (usually) the liver or kidney.
- These nanoparticles that have been completely synthesized and practically nothing is known outside the pharmaceutical industry about their structure or possible short term and long term toxicities. One justified concern is that autoimmune diseases will appear.
- Applying for a patent, the company or researcher in question must be able to proof the benefits, etc. But by submitting a patent application, all information which is new stays secret till a patent has been issued (or not). This is to protect the patent applicant against stealing sensitive data by competing pharmaceutical companies.
- Why is the nose suddenly so popular to deliver medications and nanoparticles to the brain? The barrier between the roof of the nose and the brain is very thin (ca. 5 mm) bony tissue. It is part of the basis of the skull (see figures in the text) and the olfactory nerves are multiple and penetrate through these minute openings the lamina cribrosa.
Dr. Robert Gorter has studied the current scientific literature and made a summary of what he found. And the reader will find references to scientific publications at the end.
A Patent Review on Nanotechnology-Based Nose-to-Brain Drug Delivery
(Molecular Trojan Horses)
Singh R, Brumlik C, Vaidya M, Choudhury A. A Patent Review on Nanotechnology-Based Nose-to-Brain Drug Delivery. Recent Pat Nanotechnol. 2020;14(3):174-192. doi: 10.2174/1872210514666200508121050. PMID: 32384043.
Abstract
Background: Current cerebral drug delivery to the brain and Cerebrospinal Fluid (CSF) is limited by the Blood-Brain Barrier (BBB) or the blood-blood Cerebrospinal Fluid (CSF) barrier. The popular, non-invasive, intranasal delivery provides an exciting route for topical and systemic applications. For example, intranasal drug delivery of Central Nervous System (CNS) drugs can be designed to pass the BBB barrier via the nose-to-brain pathways. Recent nanotechnology research and patenting focus mainly on overcoming typical limitations including bioavailability, transport, BBB penetration, targeted delivery, controlled release rate and controlled degradation.
Objective: The aim of the present study was to assess the state-of-the-art of nose-to-brain drug delivery systems and the role of nanotechnology in targeted delivery for the treatment of CNS and related therapeutic conditions.
Methods: Patent and related searches were made with analytics to explore and organize nanotech work in intranasal drug delivery to the brain. Technical advancements were mapped by API, formulation and performance criteria. Patents and published patent applications were searched with concept tables of keywords, metadata (e.g., assignee) and patent classes (e.g., International Patent Classes and Cooperative Patent Classes).read more
Results: The reviewed patents and published applications show a focus on formulations and therapeutic indications related to the nano-based nose-to-brain drug delivery. The main patented materials were surface modifiers, delivery systems and excipients based on nanotechnology.
Conclusion: Surface modified nanoparticles can greatly improve drug transport and bioavailability of drugs, particularly higher molecular weight drugs. The most commonly used surface modifiers were chitosan, lectin and cyclodextrin-cross-linker complex.
Nano formulations of herbal drugs could increase drug bioavailability and reduce toxicity. Biotechnology-related drug delivery approaches such as monoclonal antibodies and genetically engineered proteins (molecular Trojan horses) deliver large molecule therapeutics.
Keywords: Blood-Brain Barrier (BBB); Central Nervous System (CNS); Nanotechnology; intranasal drug delivery; neurotherapeutics; nose-to-brain.
For any queries, please email at epub@benthamscience.net
Direct nose to brain drug delivery via integrated nerve pathways bypassing the blood-brain barrier: an excellent platform for brain targeting
Pardeshi CV, Belgamwar VS. Direct nose to brain drug delivery via integrated nerve pathways bypassing the blood-brain barrier: an excellent platform for brain targeting. Expert Opin Drug Deliv. 2013 Jul;10(7):957-72. doi: 10.1517/17425247.2013.790887. Epub 2013 Apr 16. PMID: 23586809.
Abstract
Introduction: The blood-brain barrier (BBB) represents a stringent barrier for delivery of neurotherapeutics in vivo. An attempt to overcome this barrier is represented by the direct transport of drugs from the nose to the brain along the olfactory and trigeminal nerve pathways with the help of modern nanotechnology. These nerve pathways initiate in the nasal cavity at olfactory neuroepithelium and terminate in the brain. An enormous range of neurotherapeutics, both macromolecules and low molecular weight drugs, can be delivered to the central nervous system (CNS) via this route.
Areas covered: Present review highlights the literature on the anatomy-physiology of the nasal cavity, pathways and mechanisms of neurotherapeutic transport across nasal epithelium and their biofate and various strategies to enhance direct nose to brain drug delivery. The authors also emphasize a variety of drug molecules and carrier systems delivered via this route for treating CNS disorders. Patents related to direct nose to brain drug delivery systems have also been listed.
Expert opinion: Direct nose to brain drug delivery system is a practical, safe, non-invasive and convenient form of formulation strategy and could be viewed as an excellent alternative approach to conventional dosage forms. Existence of a direct transport route from the nasal cavity to the brain, bypassing the BBB, would offer an exciting mode of delivering neuro therapeutic agents; and modern nanotechnology makes this possible.
Nanoparticles for direct nose-to-brain delivery of drugs
Mistry A, Stolnik S, Illum L. Nanoparticles for direct nose-to-brain delivery of drugs. Int J Pharm. 2009 Sep 8;379(1):146-57. doi: 10.1016 / j.ijpharm.2009.06.019. Epub 2009 Jun 23. PMID: 19555750.
Abstract
This review aims to evaluate the evidence for the existence of a direct nose-to-brain delivery route for nanoparticles administered to the nasal cavity and transported via the olfactory epithelium and/or via the trigeminal nerves directly to the CNS. This is relevant in the field of drug delivery as well as for new developments in nanotechnology. Experiments in animal and human models have shown that nano-sized drug delivery systems (like liposomes) can enhance nose-to-brain delivery of drugs compared to equivalent drug solutions formulations. Protection of the drug from degradation and/or efflux back into the nasal cavity may partly be the reason for this effect of nanoparticles. It is uncertain, however, whether drug from the nanoparticles is being released in the nasal cavity or the nanoparticles carrying the drug are transported via the olfactory system or the trigeminal nerves directly into the CNS where the drug is released. Furthermore, toxicity of nanoparticulate drug delivery systems in the nasal cavity and/or in the CNS has not been extensively studied and needs to be considered carefully.
Nose-to-brain drug delivery: An update on clinical challenges and progress towards approval of anti-Alzheimer drugs
Agrawal M, Saraf S, Saraf S, Antimisiaris SG, Chougule MB, Shoyele SA, Alexander A. Nose-to-brain drug delivery: An update on clinical challenges and progress towards approval of anti-Alzheimer drugs. J Control Release. 2018 Jul 10;281:139-177. doi: 10.1016/j.jconrel.2018.05.011. Epub 2018 May 24. PMID: 29772289.
Abstract
According to the Alzheimer Association Report (2017), Alzheimer’s disease (AD) is the 6th primary cause of death in the USA, which affects nearly 5.5 million people. In the year 2017 itself, the cost of AD treatment in the USA has been reported to rise to $259 billion. This statistic shows the severity of the disease in the USA which is very much similar across the globe. On the other hand, the treatment remains limited to a few conventional oral medications (approved by FDA). These are mainly acting superficially from mild to the moderate AD. The therapeutic efficacy of the drug is not only affected by its reduced concentration in the brain owing to the existence of blood-brain-barrier (BBB) but also due to its low brain permeability. In this context, the intranasal (IN) route of drug administration has emerged as an alternative route over the systemic (oral and parenteral) drug delivery to the brain. The delivery of the drug via an IN route offers various advantages over systemic drug delivery system, as it directly delivers the drug into the brain via olfactory route. Presence of drug in the olfactory bulb, in turn, increases the drug bioavailability in the brain and reduces the drug degradation as well as wastage of the drug through` systemic clearance. However, there is also some limitation associated with IN like poor drug permeation through the nasal mucosa and mucociliary clearance. The delivery system various through novel strategies (nano drug carrier system, colloidal carriers, mucoadhesive devices, controlled delivery system, pro-drug, etc.) are adapted to overcome the above-stated limitations. Although, after all, such successful research claims, very few of the nose-to-brain drug delivery of anti-AD drugs have gained market approval due to lack of sufficient clinical evidence. Onzetra Xsail® is one such marketed preparations approved for IN delivery used for the treatment of a brain disorder; migraine. In the field of patents also, no work is found which could present sufficient experimental findings to support its clinical safety profile. It also underlines the fact that majority of work related to the nose-to-brain delivery of anti-AD drugs is limited only up to preclinical studies. In this review article, we have discussed the latest works on various novel formulations loaded with various anti-Alzheimer agents. These agents include galantamine, deferoxamine, tacrine, tarenflurbil, rivastigmine, risperidone, curcumin, quercetin, piperine, insulin, etc. and various peptides towards the development of a promising IN drug delivery system for the treatment of AD. Through this review article, we want to drag the attention of the researchers working in this field towards the challenges and hurdles of practical applicability IN delivery of anti-AD drugs. Moreover, the attention towards the clinical studies will ease the approval process for the administration of anti-Alzheimer drugs via IN route.
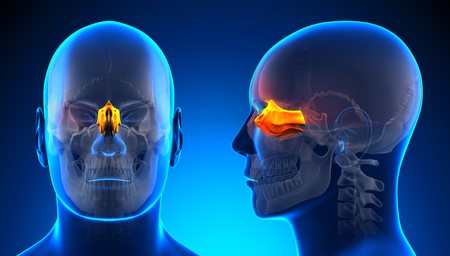
The olfactory nerve (CN I) is the first and shortest cranial nerve. It is a special visceral afferent nerve, which transmits any information relating to smell.
Embryologicallly, the olfactory nerve is derived from the olfactory placode (a thickening of the ectoderm layer), which also give rise to the glial cells which support the nerve.
In this paragraph, we shall look at the anatomy of the olfactory nerve – its structure, anatomical course and clinical relevance. And why one can deliver medications and nanoparticles through the thinnest part of the skull.
In mammalian anatomy, the cribriform plate, horizontal lamina or lamina cribrosa (from Latin cribrum, “sieve” + -form) of the ethmoid bone is received into the ethmoidal notch of the frontal bone and roofs in the nasal cavities.
The cribriform plate is narrow with deep grooves supporting the olfactory bulb, and is perforated by olfactory foramina allowing the passage of the thousands of olfactory nerve fibers.
A fractured cribriform plate can result in olfactory dysfunction, septal hematoma, cerebrospinal fluid rhinorrhoea (CSF rhinorrhoea), and possibly infection which can lead to meningitis. CSF rhinorrhoea (clear fluid leaking from the nose) is very serious and considered a medical emergency. Aging can cause the openings in the cribriform plate to close, pinching olfactory nerve fibers. A reduction in olfactory receptors, loss of blood flow, and thick nasal mucus can also cause an impaired sense of smell.
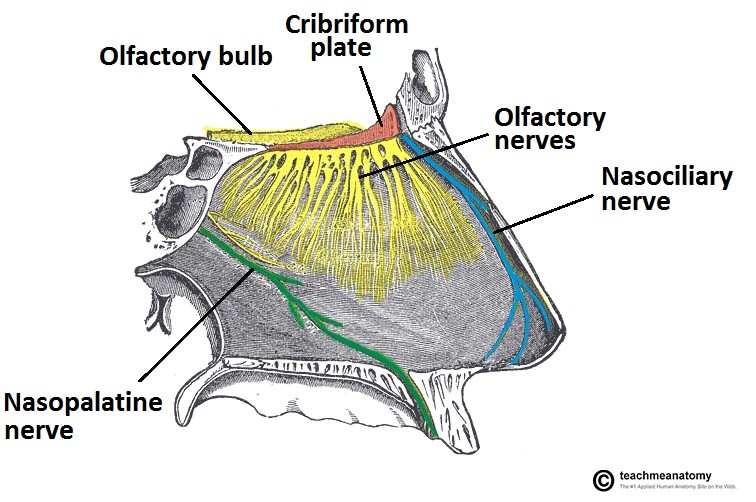
Innervation of the nasal cavity. The olfactory nerve is responsible for the sense of smell. The nasociliary and nasopalatine nerves provide general smell sensations
Through the nose directly into the brain: Biberach researchers are working on a novel way to apply pharmaceuticals
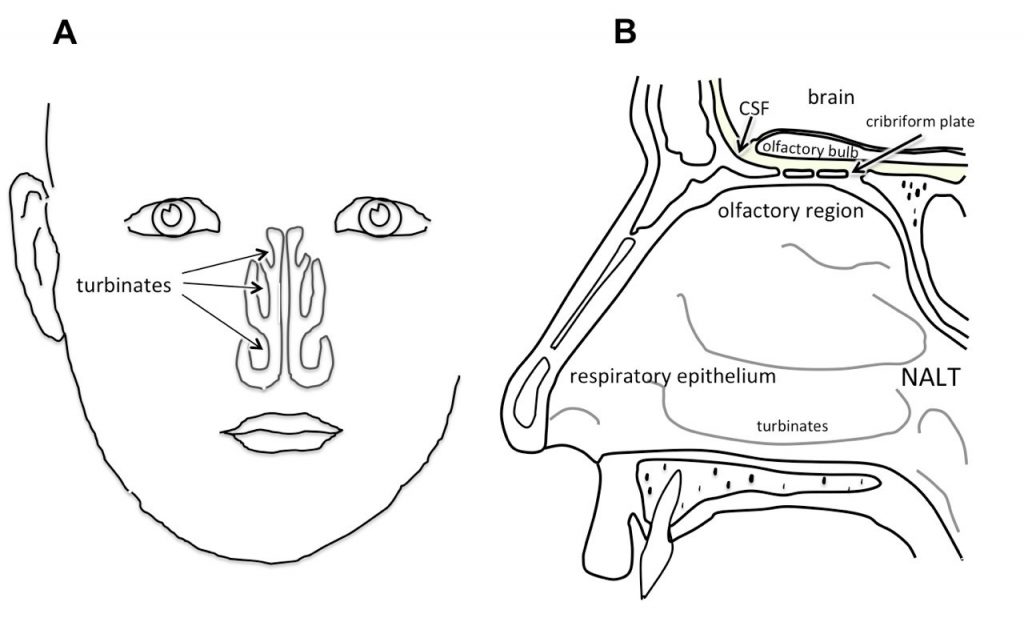
The blood-brain barrier prevents most drugs and large biologics in particular, from entering the brain. This physiological barrier impairs the study of central nervous system (CNS) diseases such as Alzheimer’s, Parkinson’s and multiple sclerosis as well as the development of drugs. However, there is a hidden side entrance to the brain, which means that there is a way to circumvent this barrier.
This hidden side entrance is through the so-called regio olfactoria (olfactory region) on the roof of the nose where the cribriform plate of the ethmoid bone and cell layers of the nasal mucosa separate the nasal cavity (outside world) from the brain (cerebrospinal fluid). The roof of the nose through which the short olfactory nerve fibers go is about 5 mm thick.
Thus; olfactory nerve fibers run through the thin with thousands of tiny canals (holes) through the cribriform plate, making this structure an excellent gateway into the brain for certain drugs, including therapeutic proteins. Moreover, drugs can also diffuse from the nose into the CNS through the trigeminal nerve.

Johannes Flamm and Prof. Katharina Zimmermann have chosen the nose as a way of administering drugs into the brain through nanoparticles as a kind of Horse of Troy.
Demonstrating technical feasibility
Researchers led by Katharina Zimmermann, professor of molecular pharmacology at the Biberach University of Applied Sciences, now want to investigate whether this route can be used to administer drugs by nanoparticles. They are also seeking to demonstrate the technical feasibility of the intranasal application of drugs, including big molecules such as therapeutic proteins. This would be an efficient, non-invasive way of administering highly specific antibodies. In addition, it would be associated with few adverse drug effects.
Johannes Flamm, funded by a Foundation of German Business grant, is a doctoral student in Prof. Zimmermann’s lab. His thesis is specifically focused on the development of a platform technology to transport proteins to their final destination by way of the CNS. The proteins he is seeking to transport must be pharmacokinetically reliable in therapeutic concentrations and currently (2021) nanotechnology can guarantee that.
Intranasal administration is undergoing thorough investigation and the research has progressed rapidly to the point that currently (2020/2021), at the Patent Bureaus in the USA and EU, Japan and other countries several patents have been applied for.
Many researchers around the world are studying how drugs can travel from the nasal mucosa into the blood stream. However, only a handful of researchers have been specifically studying the intranasal transport of drugs into the brain.
Flamm will work with two model drugs: a low-molecular weight (200 Da) muscle relaxer (baclofen) and a peptide hormone (insulin). Insulin can cross the blood-brain barrier by way of transcytosis (i.e. receptor-mediated transport of macromolecules across the interior of a cell). The hormone is an important growth and differentiation factor in the CNS and improves, as Zimmermann herself has shown, the cognitive ability of Alzheimer’s patients, at least for a short period of time. In addition, studies with Alzheimer’s patients and healthy individuals have shown that intranasally administered insulin exerts distinct influences on central nervous functions in humans. This is why the researchers from Biberach have decided to focus initially on insulin.
Nasal sprays do not work for proteins or larger molecules
Flamm and Zimmermann have been able to draw valuable conclusions from Stützle’s work: proteins that are dispersed as an aerosol are very sensitive to the shear forces inside the aerosol generator. In order to apply proteins to the nose, they need to be specifically packed (like in liposomes) or formulated. Latest developments in nanotechnology make it possible. This is not necessary for small chemical molecules, which are able to enter deep into the nasal cavity by way of gas-borne particles (aerosols) that are finely dispersed with a nasal spray. In addition, computer simulations carried out in cooperation with researchers from the University of Ulm and the Ulm University of Applied Sciences (in Germany) have shown that, when inhaled continuously, protein aerosols not only accumulate in the olfactory region as desired, but also in the nasopharynx where the mucosal immune system is located.
Pharmaceutical substances need to be protected against attack by nasal immune cells as they pass through the immunologically active areas inside the nose. In order to achieve this, Zimmermann and her team decided to work with ‘packaging specialists’ from the Stuttgart-based Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB who have developed a spray-drying procedure that enables them to load specific synthesized particles with pharmaceutical substances. Computer stimulations using standardized and idealized nose models of men and women of different ethnic origin have shown that it is possible to enclose the drug in transport vehicles with diameters of 100 or so micrometers and effectively transport them to the olfactory region. Experiments were carried out with a three-dimensional cast model of a human nasal cavity to substantiate computer simulations.
References:
Chapman, Colin D et al.: Intranasal Treatment of Central Nervous System Dysfunction in Humans. Pharmaceutical Research http://link.springer.com/article/10.1007/s11095-012-0915-1/fulltext.html
Djupesland, Per G et al.: Accessing the brain: the nose may know the way. Journal of Cerebral Blood & Flow Metabolism (2013), 33, 793-794; doi: 10.1038/jcbfm.2013.41.
Lorenzetti, Laura: Is the future of pharma about making good drugs great? Fortune, 27.2.2015.
Pardridge, William M: Drug transport across the blood-brain-barrier. Journal of Cerebral Blood Flow & Metabolism (2012), 32, 1959-1972; doi: 10.1038 / jcbfm.2012.126.
Phukan K, Nandy M, Sharma RB, Sharma HK. Nanosized Drug Delivery Systems for Direct Nose to Brain Targeting: A Review. Recent Pat Drug Deliv Formul. 2016;10(2):156-64. doi: 10.2174/1872211310666160321123936. PMID: 26996366.
Agrawal M, Saraf S, Saraf S, Antimisiaris SG, Chougule MB, Shoyele SA, Alexander A. Nose-to-brain drug delivery: An update on clinical challenges and progress towards approval of anti-Alzheimer drugs. J Control Release. 2018 Jul 10;281:139-177. doi: 10.1016/j.jconrel.2018.05.011. Epub 2018 May 24. PMID: 29772289.
Zorkina Y, Abramova O, Ushakova V, Morozova A, Zubkov E, Valikhov M, Melnikov P, Majouga A, Chekhonin V. Nano Carrier Drug Delivery Systems for the Treatment of Neuropsychiatric Disorders: Advantages and Limitations. Molecules. 2020 Nov 13;25(22):5294. doi: 10.3390/molecules25225294. PMID: 33202839; PMCID: PMC7697162.
Tavanti F, Pedone A, Menziani MC. Disclosing the Interaction of Gold Nanoparticles with Aβ(1-40) Monomers through Replica Exchange Molecular Dynamics Simulations. Int J Mol Sci. 2020 Dec 22;22(1):26. doi: 10.3390 / ijms22010026. PMID: 33375086; PMCID: PMC7792802.