Cardiovascular Assessment up to One Year After COVID-19 Vaccine–Associated Myocarditis
Presented and discussed by
Prof. Dr. Med. Robert Gorter
Authors: Yu CK, Tsao S, Ng CW, Chua GT, Chan KL, Shi J, Chan YY, Ip P, Kwan MY, Cheung YF. Cardiovascular Assessment up to One Year After COVID-19 Vaccine-Associated Myocarditis. Circulation. 2023 Aug;148(5):436-439. doi: 10.1161/CIRCULATIONAHA.123.064772. Epub 2023 Jul 31. PMID: 37523760; PMCID: PMC10373639.
Myocarditis has been recognized as a complication of COVID-19 mRNA vaccinations, particularly in adolescent and young adult male patients.1 Cardiovascular sequelae after an acute episode of myocarditis, regardless of pathogenesis, remain as issue of concern. Although the reported short-term clinical trajectory after COVID-19 vaccine–associated myocarditis appears reassuring, with a resolution of cardiac symptoms and normalization of left ventricular ejection fraction (LVEF) in most of the adolescent patients,2 the long-term cardiovascular outcomes remain unclear.
We evaluated the cardiovascular outcomes at up to 1 year in adolescent patients diagnosed with COVID-19 vaccine–associated myocarditis. Their electrocardiographic, echocardiographic, and cardiac magnetic resonance (CMR) findings at diagnosis were reviewed. At the latest follow-up, clinical assessment, ECG, echocardiogram (Vivid E9, GE Medical System, Horten, Norway), and CMR (Siemens Magnetom Aera 1.5 T MRI system, Germany) were performed. The study was approved by the Institutional Review Board and the parents of subjects gave informed consent. Data that support our findings are available from the corresponding authors upon reasonable request.
Forty patients (33 male) were 15.1±1.6 (range, 12.7–17.9) years of age and followed up for 10.0±1.3 (range, 5.6–12.3) months, representing all cases identified in Hong Kong during the period of study, were included. Twenty-nine (73%) patients were asymptomatic, whereas 7 (18%) reported non-cardiac chest pain, 3 (8%) reported palpitations, and 1 (3%) reported fatigue during follow-up, with none having cardiac arrhythmias, angina, or heart failure. The ECG results were abnormal, with ST-segment or T-wave abnormalities in 31 (78%) patients at presentation and normalized in all at the latest follow-up.
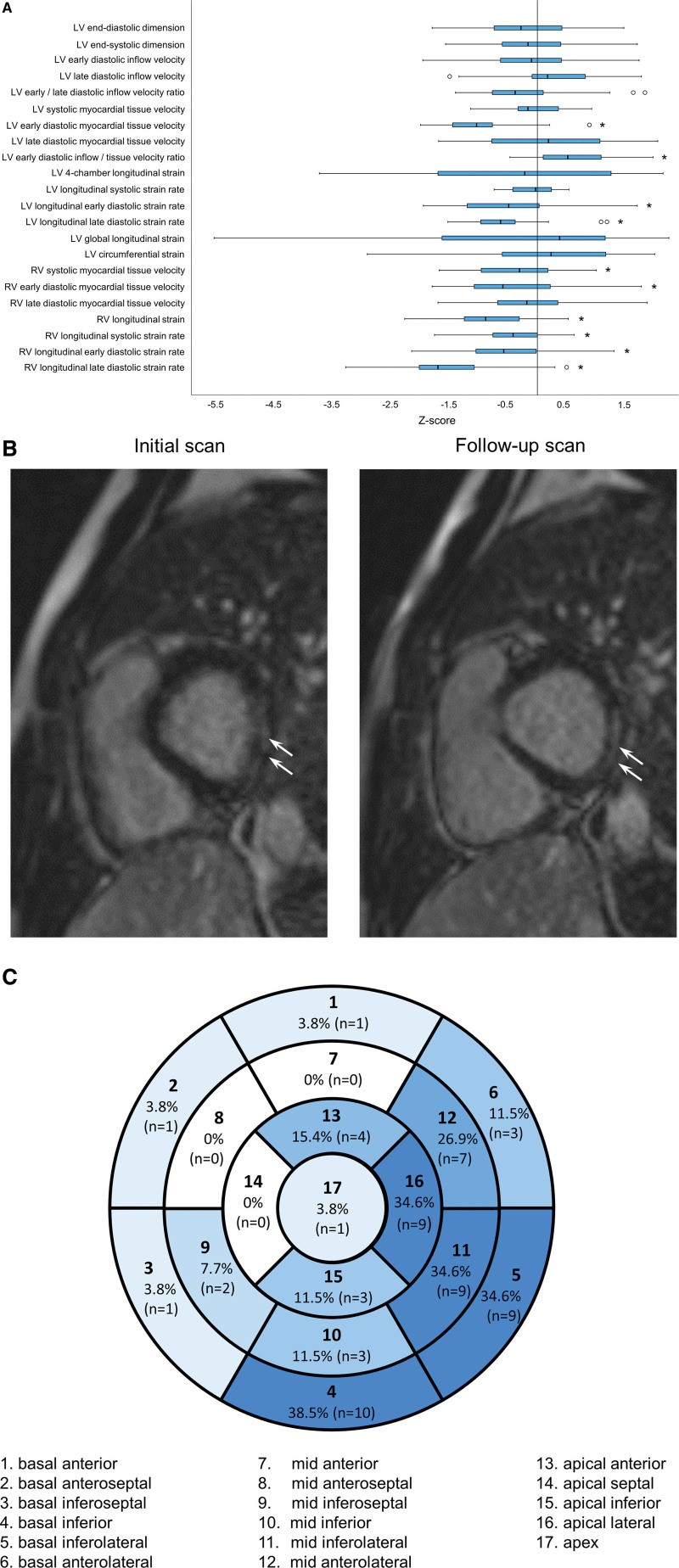
Echocardiographic assessment at presentation showed normal LVEF in all patients, mildly increased echogenicity of the pericardium or left ventricular (LV) lateral wall in 6 (15%), and minimal (<2 mm) pericardial effusion in 2 (5%). At the latest follow-up, 38 (95%) patients had normal LVEF, 2 (5%) had borderline LVEF of 51.1% and 53.6%, and all had normal right ventricular (RV) fractional area change. Analyses of RV and LV 4-chamber longitudinal strain were feasible when ≥5 segments in each plane could be analyzed, in 36 patients and LV global longitudinal and circumferential strain in 35. The LV 4-chamber longitudinal, global longitudinal, and circumferential systolic strain was impaired (Z score <–2) in 8 (22%), 7 (20%), and 4 (11%) patients, respectively. The RV longitudinal systolic strain and early- and late-diastolic strain rates were impaired in 2 (6%), 3 (8%), and 10 (28%) patients, respectively. Mean Z scores of diastolic functional indices of both ventricles were significantly lower than zero (all P<0.002; Figure A). No patients had coronary abnormalities.
Echocardiographic indices and cardiac magnetic resonance late gadolinium enhancement in adolescents at up to 1-year after COVID-19 vaccine–associated myocarditis. A, Boxplots showing the distribution of Z scores, which indicate the number of standard deviations in relation to the mean, of echocardiographic parameters (*significantly different from zero after Bonferroni adjustment for multiple comparisons). The plots illustrate the median (horizontal line), the interquartile range (IQR; box) differences in Z score between the study population and a healthy reference population, and outliers (circles) that are less than first quartile minus 1.5*IQR or greater than third quartile plus 1.5*IQR. B, Cardiac magnetic resonance images showing persistence of late gadolinium enhancement, which was assessed qualitatively using the single-shot short axis, single-shot 4-chamber stack, segmented short-axis stack, and 3-dimensional inversion recovery sequence, in the basal inferolateral segment (arrows). C, Distribution of cardiac magnetic resonance late gadolinium enhancement based on the 17-segment model. LV indicates left ventricular; and RV, right ventricular.
Thirty-nine patients had CMR at presentation, 26 of whom had abnormal findings. These included features of myocarditis in 22 (56%), abnormal T1 values in 21 (54%), abnormal T2 values in 24 (62%), late gadolinium enhancement (LGE) in 19 (49%), and reduced LVEF in 7 (18%). The CMR-derived LVEF at initial assessment correlated with LV 4-chamber longitudinal (r=0.36, P=0.04), global longitudinal (r=0.41, P=0.02), and circumferential (r=0.38, P=0.03) absolute systolic strain at follow-up echocardiography. Follow-up CMR performed in 26 patients with initial abnormal CMR findings revealed mild residual LGE in 15 (58%; Figure B), borderline LVEF in 2 (8%), and normal findings in 11 (42%) patients. None of these patients had abnormal T1 (pregadolinium 1013±28 ms and postgadolinium 473±43 ms) or T2 (48±2 ms) values or features of myocarditis. For the patient without initial assessment, follow-up CMR showed mild LGE with normal LVEF. The distribution of LGE in the 16 patients shows a predilection of involvement of lateral segments (Figure C). The presence of previous LGE correlated with LGE on follow-up (φ-coefficient=0.71, P<0.001). Patients with (n=16) and those without (n=11) LGE on follow-up CMR had similar demographic and echocardiographic parameters (all P>0.05).
To our knowledge, this is the longest follow-up study to date with comprehensive cardiac evaluation and imaging of adolescent patients diagnosed with COVID-19 vaccine–associated myocarditis. The global systolic ventricular function appears to be preserved. However, impairment of LV and RV myocardial deformation and persistence of LGE in a significant subset of patients with up to 1 year of follow-up was observed. Growing evidence suggests a worse prognosis in the presence of altered myocardial deformation and LGE in patients with myocarditis.3 A short-term study reported abnormal CMR-derived LV longitudinal strain and LGE in 70% to 75% of adolescent patients with COVID-19 vaccine–associated myocarditis followed up for 3 to 8 months.4 Similar CMR findings were found in young adults studied at 3 to 6 months after the initial diagnosis.5 The implications of impaired systolic and diastolic myocardial deformation and LGE in our patient subset remain unclear. Nonetheless, given that impaired myocardial deformation and LGE are indicators of subclinical myocardial dysfunction and fibrosis, there exists a potential long-term effect on exercise capacity and cardiac functional reserve during stress.
Limitations of our study include a relatively small patient cohort, the lack of a trajectory that depicts changes in myocardial deformation over time, and the performance of CMR only in patients with initial abnormal CMR findings. Further long-term studies to define the clinical and functional implications of these subclinical abnormalities in a larger cohort are undoubtedly warranted.
REFERENCES
- Mevorach D, Anis E, Cedar N, Bromberg M, Haas EJ, Nadir E, Olsha-Castell S, Arad D, Hasin T, Levi N, et al.. Myocarditis after BNT162b2 mRNA vaccine against COVID-19 in Israel. N Engl J Med. 2021;385:2140–2149. doi: 10.1056/NEJMoa2109730
- Kracalik I, Oster ME, Broder KR, Cortese MM, Glover M, Shields K, Creech CB, Romanson B, Novosad S, Soslow J, et al.; Myocarditis Outcomes After mRNA COVID-19 Vaccination Investigators and the CDC COVID-19 Response Team. Outcomes at least 90 days since onset of myocarditis after mRNA COVID-19 vaccination in adolescents and young adults in the USA: a follow-up surveillance study. Lancet Child Adolesc Health. 2022;6:788–798. doi: 10.1016/S2352-4642(22)00244-9
- Eichhorn C, Greulich S, Bucciarelli-Ducci C, Sznitman R, Kwong RY, Grän C. Multiparametric cardiovascular magnetic resonance approach in diagnosing, monitoring, and prognostication of myocarditis. JACC Cardiovasc Imaging. 2022;15:1325–1338. doi: 10.1016/j.jcmg.2021.11.017
- Schauer J, Buddhe S, Gulhane A, Sagiv E, Studer M, Colyer J, Chikkabyrappa SM, Law Y, Postman MA. Persistent cardiac magnetic resonance imaging findings in a cohort of adolescents with post-coronavirus disease 2019 mRNA vaccine myopericarditis. J Pediatr. 2022;245:233–237. doi: 10.1016/j.jpeds.2022.03.032
- Patel YR, Shah NR, Lombardi K, Agarwal S, Salber G, Patel R, Poppas A, Atalay MK. Follow-up cardiovascular magnetic resonance findings in patients with COVID-19 vaccination-associated acute myocarditis. JACC Cardiovasc Imaging. 2022;15:2007–2010. doi: 10.1016/j.jcmg.2022.06.009
Liu, J., Wang, J., Xu, J. et al. Comprehensive investigations revealed consistent pathophysiological alterations after vaccination with COVID-19 vaccines. Cell Discov 7, 99 (2021).
http://robert-gorter.info/superparamagnetic-nanoparticle-delivery-dna-rna-based-vaccines/
http://robert-gorter.info/covid-19-rna-based-vaccines-risk-prion-disease/
http://robert-gorter.info/covid-19-vaccines-give-people-aids-immune-system-drops-ca-5-every-week/
http://robert-gorter.info/temporary-body-storage-service/
Dr. Robert Gorter: rapidly increasing credible and peer-reviewed literature appears which documents the decline of immune function of approx. 5% per week. It can be best compared with HIV/AIDS patients whose immunocompetence declined till the patient succumbs to opportunistic infections and malignancies.